biontech
-
Pfizer’s Covid-19 Vaccine Looks Safe and Effective in Children Ages 5 to 11, Company Says
On Monday morning, pharmaceutical companies Pfizer and BioNTech announced the results of a clinical trial testing out their covid-19 vaccine in children between the ages of 5 and 11. They claim to have found that the vaccine was safe and generated a robust immune response in participants, hopefully signalling its effectiveness. The findings have yet…
-
White House May Be Clashing With CDC and FDA Experts Over Covid-19 Booster Shots
There appears to be growing tension between the Biden administration and at least some people affiliated with public health agencies over recommending covid-19 booster shots for all next month. Two senior officials with the U.S. Food and Drug Administration are leaving, reportedly in part due to the White House’s decision to promise boosters to the…
-
Pfizer Vaccine Is First to Get Full FDA Approval, Has a New Name: ‘Comirnaty’
On Monday, the U.S. Food and Drug Administration gave its full approval to Pfizer and BioNTech’s covid-19 vaccine, which has until now been covered under an emergency use authorisation. It’s now the first of its kind approved in the U.S. — a decision that public health experts say could improve its acceptance among those still…
-
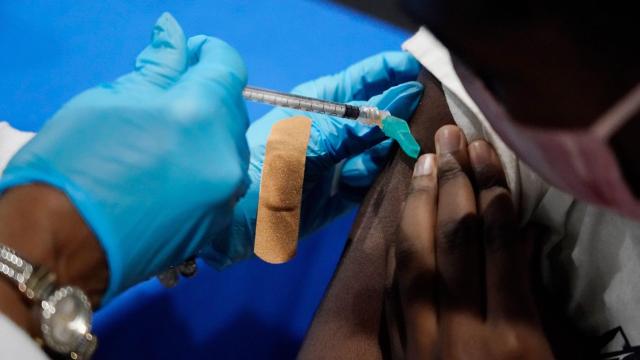
Certain Immunocompromised People Can Now Get Third Vaccine Doses in the U.S.
On Thursday, the U.S. Food and Drug Administration announced that it has authorised third Pfizer and Moderna shots for certain immunocompromised people, particularly organ transplant recipients and people diagnosed with conditions with “an equivalent level of immunocompromise.” This comes days after a randomised trial in Canada confirmed that, among 120 patients, three doses significantly boosted…